Colpidium
Colpidium Stein, 1860 (ref. ID; 2014, 7733)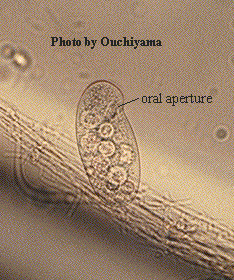
Class Oligohymenophora: Tetrahymenina (ref. ID; 5940)
Family Turaniellidae Didier, 1971 (ref. ID; 7733) [ref. ID; 2014]
Outline-shape reniform, ventral side concave, dorsal surface convex. Oral aperture located about quarter of way down body in the concavity of the ventral surface. Oral aperture relatively small and oval in outline. The undulating membrane on the right of the aperture may be visible and there are 3 membranelles on the left which may only be seen if impregnated with the silver. Uniform somatic ciliation with the kineties bending appearance. Pre-oral suture is 6-shaped at the anterior pole. Sometimes a tuft of short caudal cilia present. Contractile vacuole in posterior body half. Macronucleus ovoid, centrally located with an adjacent micronucleus.
Quote; Colin R. Curds, Michael A. Gates and David McL. Roberts "British and other freshwater ciliated protozoa Part II Ciliophora: Oligohymenophora and Polyhymenophora" Cambridge University Press, 1983 (ref. ID; 2014) [ref. ID; 7733]
Remarks
- [Stomatogenesis of Colpidium and Turaniella]: Iftode et al. (1969) have previously identified a number of similarities between the morphostatic stages of Colpidium and Turaniella. These include the disposition of the somatic kineties about the anterior pole, the oblique orientation of the oral cavity towards the ciliate's left, the elongate nature of the oral cavity, and the disposition of the paroral along the interior right and posterior edge of the oral cavity. Corliss (1953) pointed out a significant characteristic feature of kinety n of all Colpidium species: there is a break in the anterior portion of kinety n isolating a fragment of this kinety on the left anterior border of the oral cavity. This fragment is obvious in published Descriptions of Colpidium campylum (Fig.7 in Foissner 1978), Colpidium coloda (Fig.5D in Iftode et al. 1969; Fig.12 in Foissner 1978; Fig.19 in Martin-Gonzalez et al. 1981), Colpidium kleini (Fig.1 in Foissner 1973; Fig.11 in Foissner 1978), Colpidium truncatum (Fig.8 in Foissner 1978), and Colpidium uncinatum (Fig.1 in Fernandez-Galiano & Ruiz 1972 where it is identified as n-1). On the other hand, kinety n is not fragmented in genera in the Glaucomidae (Corliss 1971) nor, as Corliss (1953) pointed out, in other genera in the Tetrahymenidae. However, this feature does not separate Colpidium from Turaniella. Turaniella rather than having just a single fragment of kinety n to the anterior left of the oral cavity, has as many as 6 such fragments (see Fig 1 in Iftode & Versavel 1968). An explanation for the appearance of the fragments is found by studying the stomatogenesis of Colpidium and Turaniella. In both genera as the oral apparatus sinks below the surface and turns left, its developing right border cuts across kinety n in Colpidium and up to 6 kineties in Turaniella (Iftode et al. 1969). Although a similar leftward rotation occurs during stomatogenesis in Glaucoma (Peck 1974) and in Espejoia (Fryd-Versavel, Iftode & Wilbert 1974), this rotation does not result in the fragmentation of kinety n. Thus, this feature appears to uniquely separate both Colpidium and Turaniella from other tetrahymenine hymenostomes. In addition, the << scale >> ordered arrangement of the paroral dikinetids at the early stages of stomatogenesis in Turaniella have also been observed in C. colpoda (Figs.14, 15 in Martin-Gonzalez et al. 1981). (ref. ID; 7733)
- [Ultrastructure of the Oral Apparatus]: Ultrastructural characteristics have proven to be very useful in establishing and corroborating relationships among ciliate groups (Corliss 1979; Lynn 1981; de Puytrac et al. 1973). It is aleady established that there is a remarkable similarity in the ultrastructure of the somatic cortex of Colpodium and Turaniella (Didier et al. 1970; Lynn & Didier 1978). However, this somatic cortical pattern is characteristic of tetrahymenine hymenostomes in general. It is probable that somatic structures are more highly conserved than oral structures (Lynn 1979, 1981). Thus, if Colpidium and Turaniella are more closely related to each other than to other tetrahymenines, and expectation would be that some oral ultrastructural features would be shared. Colpidium apparently has a unique set of oral papillae in which the microtubules of the ribbed wall arise (Lynn & Didier 1978) but this feature is not found in Turaniella which has unique microfibrillar system probably performing a similar function (Didier et al. 1970). There are some peculiarities of the paroral dikinetids of Colpidium, notably that the more posterior dikinetids of the paroral are non-ciliated and lie beneath the cell membrane (Lynn & Didier 1978). For long stretches of the paroral in Turaniella, this also appears to be true. In some regions, the dikinetids of Turaniella lie beneath cortical alveoli. The paroral dikinetids of Turaniella appear to be very disorganized in morphostatic cells and this may explain why an organized ribbed-wall structure is different in this genus (Didier et al. 1970). Characterization of the isolated oral apparatus also reveals similarities between Colpidium and Turaniella (Iftode & Grain 1975). Its architecture could be described as predominantly microfilamentous; microfilamentous bundles of microfibrils apparently interconnect all the major organellar complexes of the oral apparatus and may provide a greater elasticity. This is in contrast to the isolated oral apparatus of genera in the Terahymenidae, such as Tetrahymena (Forer, Nilsson & Zeuthen 1970; Wolfe 1970), and Glaucomidae, such as Glaucoma, in which the organellar complexes are interconnected by microtubular structures rather than by microfilamentous ones. In Colpidium colpoda, Martin-Gonzalez et al. (1981) described a second paroral kinety (CP 2) along the posterior two-thirds of paroral kinety 1 (CP 1). We do not agree with this interpretation. Our observations on C. colpoda reveal that the entire paroral is composed of dikinetids; those in its posterior two-thirds have the oral ribs which are often visible after silver-staining. The CP 2 of Martin-Gonzalez et al. (1981) is probably the dense material of the papillae sometimes revealed by silver-staining. Their drawing (Fig.1 in Martin-Gonzalez et al. 1981) of the oral fibrillar system is partially correct: their << fibras citostomicas >> is not as prominent and not in the same place as that which is visible in our Fig. 3. This fibre runs along the right side of OPK 3, in the same position as the cytostomial fiber << en dent de peigne >> described in Turaniella (Iftode et al. 1969; Iftode & Grain 1975). But, we observed only two prominent << cordones fibrosos >> in the middle of the isolated oral apparatus: one is at the level of the eighth oral papilla and the other is at the level of the twelfth oral papilla. Thus, in addition to the gross similarity in the longitudinal orientation of the oral polykinetids in the oral cavity and the elongate linear nature of the oral polykinetids of Colpidium and Turaniella, there are two shared ultrastructural features of the oral apparatus: first, non-ciliated kinetids lie beneath the plasmlemma and sometimes beneath alveoli; second, the structural linkages between the oral organellar complexes are predominantly microfilamentous rather than microtubular. (ref. ID; 7733)
- [Transfer of Colpidium to the Turaniellidae Didier, 1971]: There is enough evidence now suggesting a close relationship between Colpidium and Turaniella to formalize this by transferring the genus Colpidium to the family Turaniellidae Didier, 1971. (ref. ID; 7733)
- Colpidium acuminatum (ref. ID; 4611)
- Colpidium campylum (Stokes) (ref. ID; 1219, 1618, 3698, 5462), (Stokes, 1886) Bresslau, 1922 (ref. ID; 1622, 1882, 2245, 3116, 4611) reported author and year? (ref. ID; 65, 191, 3954, 5940), campyllum (Stokes) Bresslau (ref. ID; 3342)
See; Dexiostoma campylum Ganner & Foissner, 1989 (ref. ID; ?) or (Stokes, 1886) Jankowski, 1967 (ref. ID; 4611)
Syn; Cryptochilum griseolum var. marium Gourret & Roeser, 1866 (ref. ID; 1622); Glaucoma colpidium Schewiakoff, 1896 (ref. ID; 1622); Tillina campylum Stokes, 1886 - Colpidium caudatum Wilbert, 1982 (ref. ID; 503 original paper)
- Colpidium colpidium (Schewiakoff) (ref. ID; 5462)
- Colpidium colpoda (ref. ID; 65, 191)
- Colpidium colpoda (Ehrenberg, 1831) (ref. ID; 3593) reported year? (ref. ID; 1618, 3698, 5462), (Ehrenberg, 1831) Stein, 1869 (ref. ID; 1219, 1335, 1622, 1629, 1896, 2245) or 1860 (ref. ID; 3116) reported year? (ref. ID; 646, 3342)
Syn; Colpidium cucullus Kent, 1882 (ref. ID; 3116); Paramecium colpoda Ehrenberg, 1831 (ref. ID; 3116); Tillina campylum Stokes, 1886 (ref. ID; 1622, 3116); Tillina helia Stokes, 1885 (ref. ID; 1622) reported year? (ref. ID; 1618) - Colpidium colpoda (Losana, 1829) Stein, 1860 (ref. ID; 4488, 4611)
Syn; Paramaecia kolpoda Losana, 1829 (ref. ID; 4611) - Colpidium cucullus Kent, 1882
See; Colpidium colpoda (ref. ID; 3116) - Colpidium echini (Russo) (ref. ID; 1618)
- Colpidium kleini Foissner, 1969 (ref. ID; 4488, 4611, 7733) reported year? (ref. ID; 3698) reported author and year? (ref. ID; 191, 1629)
- Colpidium pannonicum V. Gelei, 1932 (ref. ID; 1620)
- Colpidium singulare (ref. ID; 4611)
- Colpidium striatum Stokes, 1886 (ref. ID; 1622) reported year? (ref. ID; 1618, 5462) reported author and year? (ref. ID; 65)
- Colpidium truncatum Stokes, 1885 (ref. ID; 1622, 4611) reported year? (ref. ID; 3698, 5462) reported author and year? (ref. ID; 191)
See; Paracolpidium truncarum (ref. ID; 4611)
Colpidium campylum (Stokes) (ref. ID; 1219, 1618, 3698, 5462), (Stokes, 1886) Bresslau, 1922 (ref. ID; 1622, 1882, 2245, 3116, 4611) reported author and year? (ref. ID; 65, 191, 3954, 5940), campyllum (Stokes) Bresslau (ref. ID; 3342)
See
Dexiostoma campylum Ganner & Foissner, 1989 (ref. ID; ?) or (Stokes, 1886) Jankowski, 1967 (ref. ID; 4611)Synonym
Cryptochilum griseolum var. marium Gourret & Roeser, 1866 (ref. ID; 1622); Glaucoma colpidium Schewiakoff, 1896 (ref. ID; 1622); Tillina campylum Stokes, 1886Descriptionss
Elongated, reniform, sometimes ovoid; uniform ciliation except for a group of longer cilia at the posterior pole; preoral suture slightly displace towards the ventral side; suture curved to right; small triangular buccal cavity near the right side of the body at about 1/4 of the body length from the anterior pole; buccal ciliation tetrahymenal; 1 undulating membrane on the right and 3 adoral membranelles on the left; 1 spherical macronucleus and 1 micronucleus in the central part of the body; 1 contractile vacuole located near the right margin. (ref. ID; 1219)Elongate reniform; 27-30 ciliary meridians; preoral suture curved to right; in fresh and brackish water. (ref. ID; 1618)
Measurements
Length 50-120 um. (ref. ID; 1219)50-70 um long. (ref. ID; 1618)
45-60x20-25 um. (ref. ID; 3342)
Colpidium colpoda (Ehrenberg, 1831) (ref. ID; 3593) reported year? (ref. ID; 1618, 3698, 5462), (Ehrenberg, 1831) Stein, 1869 (ref. ID; 1219, 1335, 1622, 1629, 1896, 2245) or 1860 (ref. ID; 3116) reported year? (ref. ID; 646, 3342)
Synonym
Colpidium cucullus Kent, 1882 (ref. ID; 3116); Paramecium colpoda Ehrenberg, 1831 (ref. ID; 3116); Tillina campylum Stokes, 1886 (ref. ID; 1622, 3116); Tillina helia Stokes, 1885 (ref. ID; 1622) reported year? (ref. ID; 1618)Descriptions
Ovoid or reniform, elongated; except for a few long caudal cilia, uniform ciliation about 55 ciliary meridians; preoral suture curved to the left and widely displaced to the ventral side; buccal cavity near the right side of the body at about one-quarter of the body length from the anterior pole; buccal ciliation similar to that of the C. campylum; 1 spherical macronucleus, a single micronucleus; the contractile vacuole is located in the central part of the body near the dorsal face. (ref. ID; 1219)Elongate reniform; cytostome about one-tenth the body length; 55-60 ciliary meridians; preoral suture curved to left; macronucleus oval, central; a micronucleus; fresh water. (ref. ID; 1618)
Measurements
Length 100-150 um. (ref. ID; 1219)90-150 um long. (ref. ID; 1618)
70-80x34-44 um. (ref. ID; 3342)
Colpidium echini (Russo) (ref. ID; 1618)
Descriptions
In the intestinal caeca of Strongylocentrotus lividus; 24 longitudinal ciliary rows; cytostome at anterior third. (ref. ID; 1618)Measurements
37-64 (55) by 21-28 (25) um. (ref. ID; 1618)Colpidium kleini Foissner, 1969 (ref. ID; 4488, 4611, 7733) reported year? (ref. ID; 3698) reported author and year? (ref. ID; 191, 1629)
Descriptions
Colpidium kleini measures about 90 um in length by about 30 um in width after protargol staining. It possesses about 37 somatic kineties of which 2 are postoral. The cytoproct is posterior and placed between these two postoral kineties. At the anterior pole, the right somatic kineties curve anterior to the oral region. The left somatic kineties converge on these right kineties and form a long, oblique anterior suture. About a dozen of the dorsal somatic kineties terminate with a dikinetid. The contractile vacuole pore is located at the posterior end of kinety 8. A cortical feature of key importance is the kinetal fragment just to the left of the oral region. This fragment appears to be a anterior portion of kinety n which has become separated during stomatogenesis. This interpretation is supported by a stomategenetic analysis. The oral infraciliature is composed of a paroral of dikinetids and three polykinetids. The polykinetids differ in structure as follows: OPK 1 has two rows of 24 kinetosomes and a left-most one of 6 kinetosomes; OPK 2 has three rows of 26 kinetosomes; and OPK 3 has 4 rows of decreasing length with the left-most having 26 kinetosomes while the right-most, deep in the oral cavity, has only 12 kinetosomes. The cytopharyngeal fibre is very long. (ref. ID; 7733)- [Stomatogenesis]: Stomategensis begins with the appearance of kinetosomal replication equatorially along somatic kinety 1. This anlagen rapidly takes an elongate shape with the kinetosomes aligning in linear, longitudinal fashion at the time micronuclear mitosis begins; this field is dissimilar to the anarchic field of Tetrahymena. The polykinetids are initially composed of only two files of kinetosomes and precede the formation of the paroral membrane. At this time, the somatic kinetosomes begin to replicate in the equatorial region. To this stage, the oral anlagen is superficial; the micronucleus has not yet completely divided. When the fission furrow begins to form, the posterior end of the opisthe oral anlagen begins to sink below the cell surface; the anterior end follows to form an oral cavity. Just prior to this, dikinetids have aligned along the right edge of the anlage to form the paroral membrane. The polykinetids at this time have a full complement of 3 or 4 kinetosomal files. There is some oral reoganisation in the proter whose cytopharyngeal fibre regresses; a new contractile vacuole pore for the proter forms at this time. At last, the polykinetids and the paroral of the opisthe assume their fully differentiated positions with the appearance of the cytopharyngeal fibres of the opisthe. A feature of key importance during stomatogenesis of Colpidium is the cortactile remodelling that occurs at this stage. The oral anlage rotates and segregates the anterior segment of kinety n which becomes a small kinetal fragment to the left of the oral region. The macronucleus and micronucleus complete division. The right somatic kineties of the opisthe begin to incline towards the left and turn around the anterior end. It is at the moment of separation of the cells that dikinetids appear at the anterior ends of the dorsal somatic kineties of the opisthe. (ref. ID; 7733)
- [The Oral Apparatus]: The isolated oral apparatus of Colpidium is held together by a network of microfilamentous material a has been described previously by Lynn and Didier (1978). OPK1 and OPK2 are initimately joined together by filamentous material and postciliary fibres while OPK 2 and OPK 3 are indirectly joined by a filamentous strand that appears to extend longitudinally beneath a ridge which rum between these two polykinetids. Smaller microfilamentous strands extend from this longitudinal bundle towards the kinetosomes of both OPK 2 and OPK 3 and the postciliary ribbons of OPK 2. Only the anterior six to eight paroral dikinetids have a ciliated anterior kinetosome. In the isolated oral apparatus, the oral ribs appear as a series of curved, densely stained structures which usually overlie the polykinetids. In some preparations where this overlying does not occur, the dense papillae in which the oral rib microtubules arise are visible. These papillae extend over the non-ciliated paroral dikinetids of the posterior part of the paroral. The paroral dikinetids are underlain by microfilaments and microtubules. The anterior kinetosome has a parasomal sac on its right; the posterior kinetosome has a small group of somatic postciliary microtubules. The dikinetid kinetosomes are linked to each other by dense, perhaps periodic, material. The right and left components of the oral apparatus are joined by two transverse strands termed << cordones fibrosos >> by Martin-Gonzalez et al. (1981). Our electron micrographs of sectioned cells suggest that these fibres are composed of both microtubules and microfilaments. The microtubular fibres of the cytopharynx isolated with the oral apparatus originate from the oral ribs of the paroral lip and not from the polykinetids. (ref. ID; 7733)